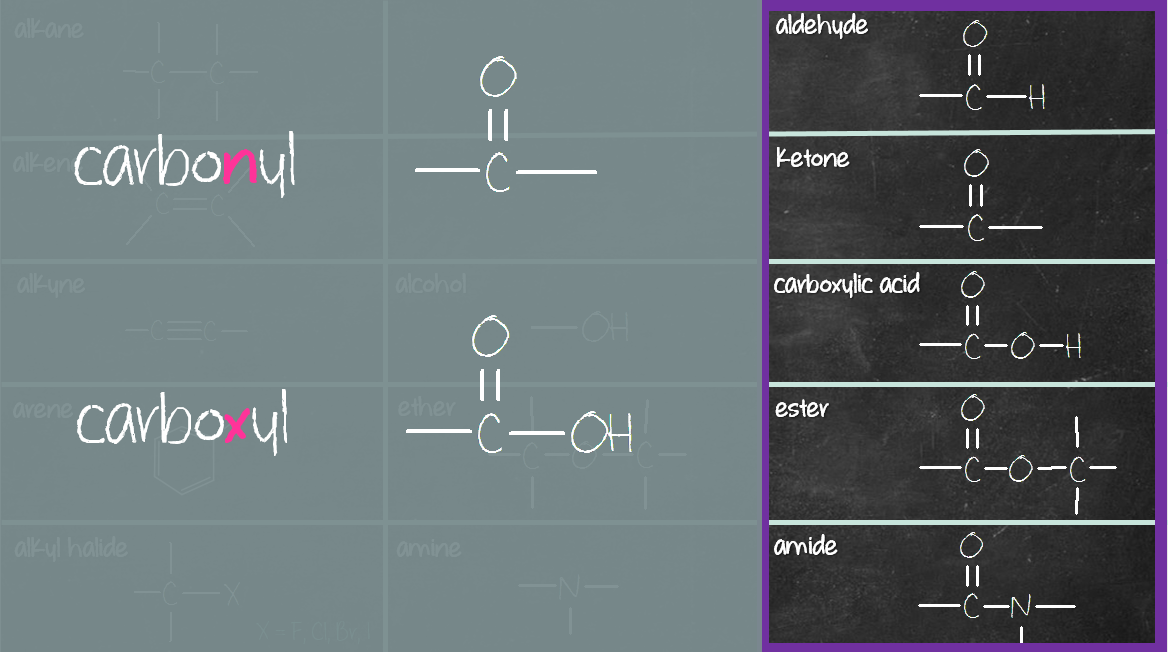
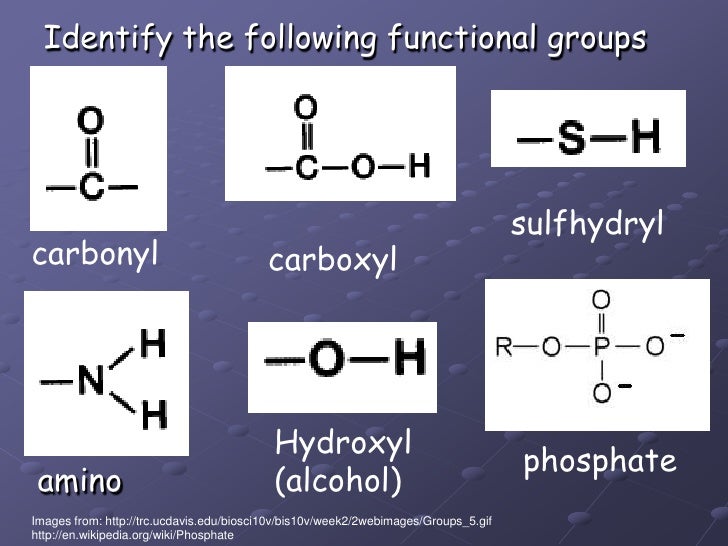
Thus, the carbonyl, C=O, and hydroxy, OH, of a carboxylic acid, RCOOH, are part of a single function and are NOT "alcohol-plus-ketone":Ī Couple of Words About The Functional Group Approach The rule is that functions assume their distinct identity when separated by –CH 2– groups. Organic molecules are also named using the functional group approach: Therefore, it is only necessary to know about the chemistry of a few generic functions in order to predict the chemical behaviour of thousands of real organic chemicals. The functional group approach " works" because the properties and reaction chemistry of a particular functional group (FG) can be remarkably independent of environment. An inert hydrocarbon skeleton onto which functional groups (FGs) are attached or superimposed.Organic chemistry is dominated by the "functional group approach", where organic molecules are deemed to be constructed from: Briefly, the sequence rules rank the substituents in order of decreasing atomic number and if two or more atoms connected to the C atom are the same the second atom determines the order. The other isomer is E (for e ntgegen meaning opposite). If the two with the higher rankings are on the same side of the double bond, that isomer is Z (for the German word zusammen, which means together). The newer method ranks the substituents for each C atom according to the Cahn-Ingold-Prelog sequence rules. But it won’t work well if there were four different atoms involved. The older method uses cis- and trans- which works well for the example given above because there are two H atoms and two Cl atoms. The isomers are diasteromers according to the definition given here. These isomers are superimposable on their mirror images if no chiral centers are present. Geometric isomers are generally not optical isomers unless they also happen to have chiral centers. Geometric isomers are stereoisomers that are distinct and separate because they cannot freely rotate due to a multiple bond or a ring structure. The bond length in C=O, for example, is greater in acetaldehyde than those in formaldehyde, which naturally takes into consideration the attractive interaction of C H 3 C C H 3 in the compound.Stereoisomers (briefly and non-optical only!)
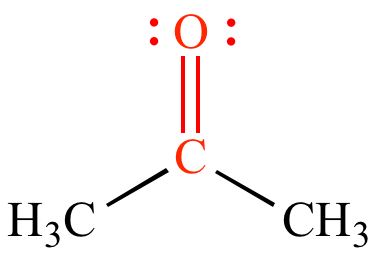
The extent of a carbonyl bond can be matched by its polarity the further the bond is lengthened, the reduced the polarity. Any double bond of a carbonyl needs to extend to approximately 1.2 angstroms and the frequency is around 176-179 kilocalorie/mol). For instance, the electronegativity of oxygen similarly polarizes the pi bond enabling the single-bonded substituent connected with becoming electron pulling out. The carbonyl group's properties are straightforwardly attached to its electronic structure as well as geometrical positioning. One combination of lone oxygen groups is located in 2s whereas the other group is in orbital 2p where its axis is directed exactly to reverse the pi orbitals' path. The C=C is less responsive because of C=O electronegativity ascribed to the oxygen and its two lone pairs of electrons. Carbonyl group double bonds: In form of reactions, the double bonding in carbonyl and the double bonding in alkenes are Really specific. Of course, different effects can assume a function in this selectivity process, including steric effects, electronic effects, and thermodynamic against kinetic reaction control. From the other half of the atom, even the more nucleophilic aldehyde interacts with the methyl ketone enolate. Two possible electrophilic positions occur in the supposedly left reactant: an ester and an aldehyde dehydrogenase. Ketones contain secondary alcohols, while primary alcohols are produced by esters, carboxylic acid, and aldehydes. By association with hydride materials such as NaBH4 and LiAlH4, with baker's yeast, or by catalytic hydrogenation, carbonyl classes may be reduced.
